In India, the accepted lower age limit for OPV is the day of birth. For IPV, the lower age limit that I would personally recommend is 8 weeks of age, since we have clearly shown the dampening effect of maternal antibody on antibody response to IPV.
Who is most at risk for polio?
Polio is still a problem in poor, undeveloped countries. Infants and children are at the greatest risk, and infections are most common during summer and fall.
Since IPV is completely safe, no such upper limit was stipulated for it even in the United States. So IPV was (and is) recommended for those who travel to polio endemic countries such as India. In previously fully immunized persons the reinforcing dose could be either OPV or IPV, since they have no risk of OPV paralysis.
Do all babies need polio drops?
CDC recommends that all children get 4 doses of polio vaccine as part of the routine childhood vaccination schedule, Adults with increased risk of exposure to poliovirus may receive one lifetime IPV booster
Let us look at OPV first. In India, virtually all cases of poliomyelitis occur below 5 years of age. This means that nearly 100 per cent of the population would have already been infected with, and hence immune to, all 3 types of polioviruses. However, on rare occasions we have seen poliomyelitis in older children and even in adults; these are exceptions which are ignored for preparing general guidelines. In other words, guidelines are based on proba-bilities of risk of disease, and the risk of disease is extremely low, indeed negligible, beyond 5 years of age. Therefore, OPV is not usually recommended beyond 5 years, either as the first dose or as a reinforcing dose. Theoretically speaking, the age range of polio might go up in well immunized communities, due to the retardation of circulation of the causative wild polioviruses. It is for this reason that the World Health Organization recommends to monitor all cases of acute flaccid paralysis in children up to 15 years and to investigate them for poliovirus etiology, for the purpose of certi-fication of polio eradication. However, all available data in India indicate that wild poliovirus isolations are almost exclusively from under-five children. Therefore, the upper age for OPV is still 5 years, either for the first dose or for the ‘umpteenth’ dose. These days, I cannot say what is the last dose since some 4-year-old children may receive up to 17-19 doses in the States in which 4 doses are being given in campaigns. In other States where 6 doses are given, the number could be as high as 19-21 doses. If such a child lives in the vicinity of another child with AFP, then one or more additional doses may also be given. For all these indications of OPV, the upper age limit is set as 5 years.
In the late 19th and early 20th centuries, frequent epidemics saw polio become the most feared disease in the world. A major outbreak in New York City in 1916 killed over 2000 people, and the worst recorded US outbreak in 1952 killed over 3000.
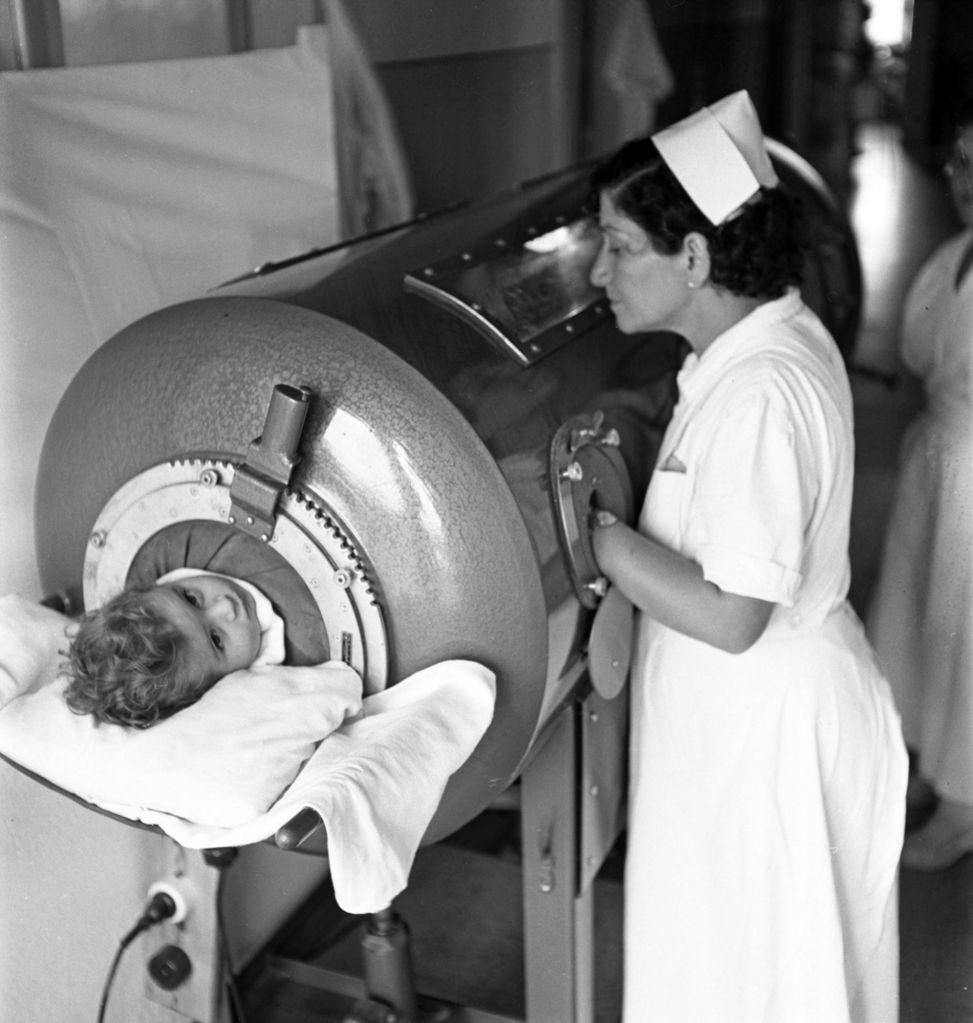
Many who survived the disease faced lifelong consequences. Deformed limbs meant they needed leg braces, crutches or wheelchairs, and some needed to use breathing devices like the iron lung, an artificial respirator invented for treatment of polio patients.
By the mid-20th century, the poliovirus could be found all over the world and killed or paralysed over half a million people every year. With no cure, and epidemics on the rise, there was an urgent need for a vaccine.
A breakthrough occurred in 1949, when poliovirus was successfully cultivated in human tissue by John Enders, Thomas Weller and Frederick Robbins at Boston Children’s Hospital. Their pioneering work was recognized with the 1954 Nobel Prize.
Not long afterwards, in the early 1950s, the first successful vaccine was created by US physician Jonas Salk. Salk tested his experimental killed-virus vaccine on himself and his family in 1953, and a year later on 1.6 million children in Canada, Finland and the USA.
The results were announced on 12 April 1955, and Salk’s inactivated polio vaccine (IPV) was licensed on the same day. By 1957, annual cases dropped from 58 000 to 5600, and by 1961, only 161 cases remained.
Salk was committed to equitable access to his vaccine, and understood that elimination efforts would not work without universal low- or no-cost vaccination.
Six pharmaceutical companies were licensed to produce IPV, and Salk did not profit from sharing the formulation or production processes.
In a 1955 interview, when asked who owned the patent for IPV, he replied: “Well, the people, I would say. There is no patent. Could you patent the sun?”
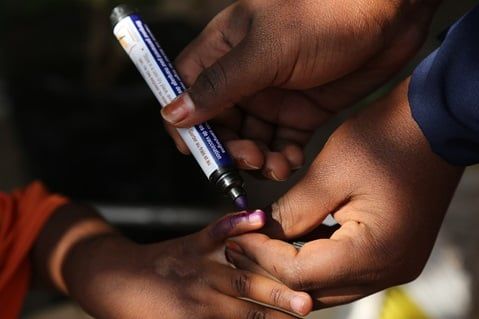
As at July 2021, only 2 cases of wild poliovirus have been recorded globally this year to date: one each in Afghanistan and Pakistan.
But alongside the success of the OPV comes a disadvantage: continued use of the vaccine poses a risk to wiping out the disease.
While OPV is safe and effective, in areas where vaccination coverage is low, the weakened vaccine virus originally contained in OPV can begin to circulate in undervaccinated communities.
When this happens, if it is allowed to circulate for sufficiently long enough time, it may genetically revert to a ‘strong’ virus, able to cause paralysis, resulting in what is known as circulating vaccine-derived polioviruses (cVDPVs).
If a population is adequately immunized, it will be protected against both wild and vaccine-derived polioviruses.
Watch this short video and learn how different strains of the polio viruses emerge and how to stop them.
Information source
T. Jacob John,
Emeritus Medical Scientist (ICMR),
Leave a comment